Suzhou, China, May 11th, 2026— CStone Pharmaceuticals (“CStone,” HKEX: 2616), an innovation-driven biopharmaceutical company focused on the research and development of therapies for oncology, immunology, inflammation, and other key disease areas, today announced the successful production and release of its first batch of domestically manufactured pralsetinib capsules (100 mg). The shipment marks the official commencement of the drug’s nationwide commercial distribution, signifying a major milestone in securing a localized supply chain for this innovative therapy.
First batches of locally-manufactured pralsetinib capsules products at the shipment site

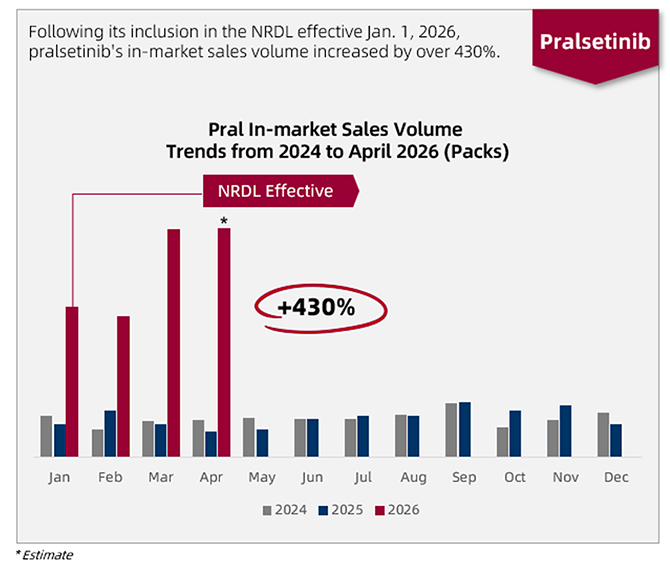
Following the inclusion of pralsetinib capsules in China’s National Reimbursement Drug List (NRDL) in late 2025—effective January 1, 2026—patient access has improved significantly. As of April 2026, in-market sales volume has surged by over 430% year-on-year. Capitalizing on this growth trajectory, CStone projects that full-year 2026 sales revenue for pralsetinib capsules will exceed RMB 300 million.
Pralsetinib capsules in-market sales volume increased by over 430%
Dr. Jason Yang, CEO, President of R&D, and Executive Director at CStone, stated, “The seamless execution of this first domestic shipment reflects the rigor of our cross-functional teams in manufacturing, quality, regulatory, and supply chain. Through stringent technical transfers and process validation, we have ensured that our locally manufactured pralsetinib capsules is highly comparable to the imported version in both quality and efficacy, fully adhering to global standards. With a mature domestic supply chain now in place, we are well-positioned to meet the surging clinical demand driven by NRDL inclusion, while also optimizing our cost structure and enhancing margin resilience. Moving forward, CStone remains steadfast in our commitment to delivering high-quality, stable drug supply to patients across China.”
About Pralsetinib Capsules
Pralsetinib capsules is a once-daily oral targeted therapy approved by the National Medical Products Administration (NMPA) of China for the first-line treatment of adults with locally advanced or metastatic RET fusion-positive NSCLC, the treatment of adults with locally advanced or metastatic RET fusion-positive NSCLC previously treated with platinum-based chemotherapy; and RET fusion-positive thyroid cancer (TC). In addition, this medicine has been approved by the Department of Health of the Government of Hong Kong (HK DoH) for the treatment of patients with RET fusion-positive locally advanced or metastatic NSCLC and it has been approved by the Taiwan Food and Drug Administration (TFDA) for the treatment of adult patients with locally advanced or metastatic RET fusion-positive NSCLC and advanced or metastatic RET fusion-positive TC.
GAVRETO® is approved by the U.S. Food and Drug Administration (FDA) for the treatment of:
*This indication is approved under accelerated approval based on Objective Response Rate (ORR) and duration of response (DOR). Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Pralsetinib capsules was discovered by CStone’s partner, Blueprint Medicines (acquired by Sanofi in July 2025). CStone has an exclusive collaboration and license agreement with Blueprint Medicines for the development and commercialization of pralsetinib capsules in Greater China, which encompasses Mainland China, Hong Kong, Macau and Taiwan. In November 2023, CStone announced an exclusive agreement with Shanghai Allist Pharmaceuticals Co., Ltd. to commercialize pralsetinib capsules in Mainland China.
About CStone
CStone (HKEX: 2616), established in late 2015, is an innovation-driven biopharmaceutical company focused on the research and development of therapies for oncology, immunology, inflammation, and other key disease areas. Dedicated to addressing patients’ unmet medical needs in China and globally, the Company has made significant strides since its inception. To date, the Company has successfully launched 4 innovative drugs and secured approvals for 21 new drug applications covering 9 indications. The company’s pipeline is balanced by 16 promising candidates, featuring antibody-drug conjugates (ADCs), multispecific antibodies, immunotherapies and precision medicines. CStone also prides itself on a management team with comprehensive experiences and capabilities that span the entire drug development spectrum, from preclinical and translational research to clinical development, drug manufacturing, business development, and commercialization.
For more information about CStone, please visit: www.cstonepharma.com.
IR contact: ir@cstonepharma.com
PR contact: pr@cstonepharma.com
Forward-looking statements
The forward-looking statements made in this article only relate to events or information as of the date when the statements are made in this article. Except as required by law, we undertake no obligation to update or publicly revise any forward-looking statements, whether as a result of new information, future events or otherwise, after the date on which the statements are made or to reflect the occurrence of unanticipated events. You should read this article completely and with the understanding that our actual future results or performance may be materially different from what we expect. All statements in this article are made on the date of publication of this article and may change due to future developments.
Disclaimer: only for communication and scientific use by medical and health professionals, it is not intended for promotional purposes.
Blueprint Medicines and associated logos are trademarks of Blueprint Medicines Corporation. GAVRETO® and associated logos are trademarks of Blueprint Medicines Corporation outside of the United States.
Your privacy is important for us. We use cookies to enhance your experience when visiting our websites: performance cookies show us how you use this website, functional cookies remember your preferences and targeting cookies help us to share content relevant to you. Select “Accept all” for giving your consent to all cookies or select “Reject all” for using only strictly necessary cookies.