Suzhou, China, April 20th, 2026—CStone Pharmaceuticals (“CStone,” HKEX: 2616), an innovation-driven biopharmaceutical company focused on the research and development of therapies for oncology, immunology, inflammation, and other key disease areas, today announced that the Company presented the latest preclinical data for three proprietary pipeline assets at the American Association for Cancer Research (AACR) Annual Meeting (from April 17 to 22), including CS5007 (EGFR/HER3 ADC), CS5006 (ITGB4 ADC), and CS5008 (DLL3/SSTR2 ADC).
CStone's Proprietary ADC Technology Platform
All three antibody-drug conjugates (ADCs) presented at AACR – CS5007 (EGFR/HER3 ADC), CS5006 (ITGB4 ADC), and CS5008 (DLL3/SSTR2 ADC) – are developed utilizing CStone’s proprietary ADC technology platform, which incorporates the following core features:
CS5007 – EGFR/HER3 Bispecific ADC
EGFR and HER3, members of the ErbB receptor family, are key oncogenic drivers frequently co-overexpressed across a variety of human epithelial malignancies. Although single-target EGFR therapies are widely utilized in standard-of-care treatments, adaptive resistance driven by compensatory HER3 signaling and heterodimerization substantially limits long-term clinical benefit. Therefore, dual targeting of EGFR and HER3 represents a highly promising strategy to overcome the tumor heterogeneity and resistance mechanisms that commonly compromise single-target approaches. CS5007 is designed to synergistically bind EGFR and HER3, which form extensive dimerization with other HER family members, thereby targeting almost all oncogenic HER-family receptor complexes (except HER2 homodimers) and effectively blocking the signaling cascades that promote tumor cell survival and proliferation.
CS5007 is a bispecific ADC comprising: 1) an anti-EGFR and HER3 human IgG1 antibody; 2) CStone’s proprietary hydrophilic CSL20 linker; 3) exatecan (Exa), a clinically validated topoisomerase I inhibitor, as the payload, conjugated with a drug-to-antibody ratio (DAR) of approximately 4.
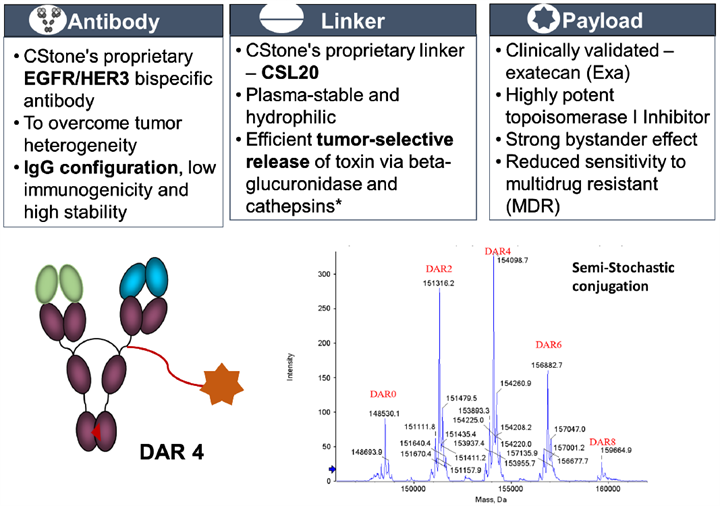
Notes: * Beta-glucuronidase exclusively functions within the cell and is highly expressed in tumor cells.
Key Highlights:
1. Superior Molecular Stability
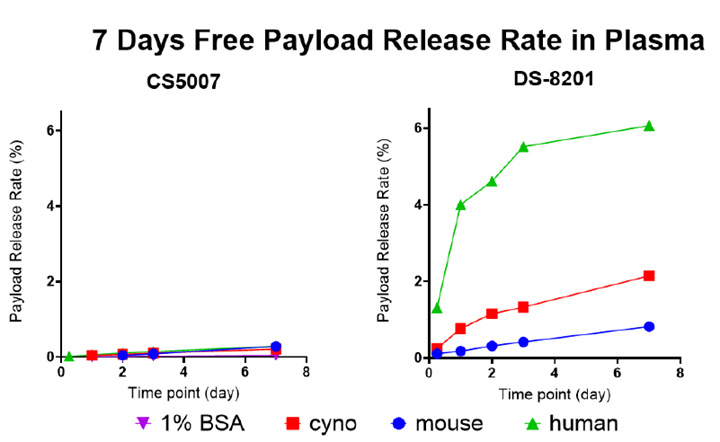
CS5007 demonstrates excellent stability in in vitro plasma stability tests, outperforming DS-8201 (trastuzumab deruxtecan) benchmark. After 7 days of incubation in plasma, free payload release was below 0.5%, indicating a low risk of off-target toxicity.
2. Dual Signaling Pathway Blockade
Western blot analysis was used to assess the signal-blocking capability of J17 (the naked antibody of CS5007) across various human tumor cell lines under a continuous stimulation microenvironment simulated by ligands (TGF-α/EGF and/or NRG1 β1). By dually targeting EGFR and HER3, CS5007 achieves potent inhibition of downstream signaling cascades, including the Akt and MAPK pathways. This dual blockade overcomes the inherent limitations and resistance mechanisms associated with single-target inhibition. Compared to SI-B001 (the naked antibody of BL-B01D1), CS5007’s antibody (J17) demonstrates superior inhibitory potency. Notably, while SI-B001 fails to interrupt HER3/Akt signaling in A431 and FaDu cells, J17 effectively abrogates these signals even under ligand-stimulated conditions.
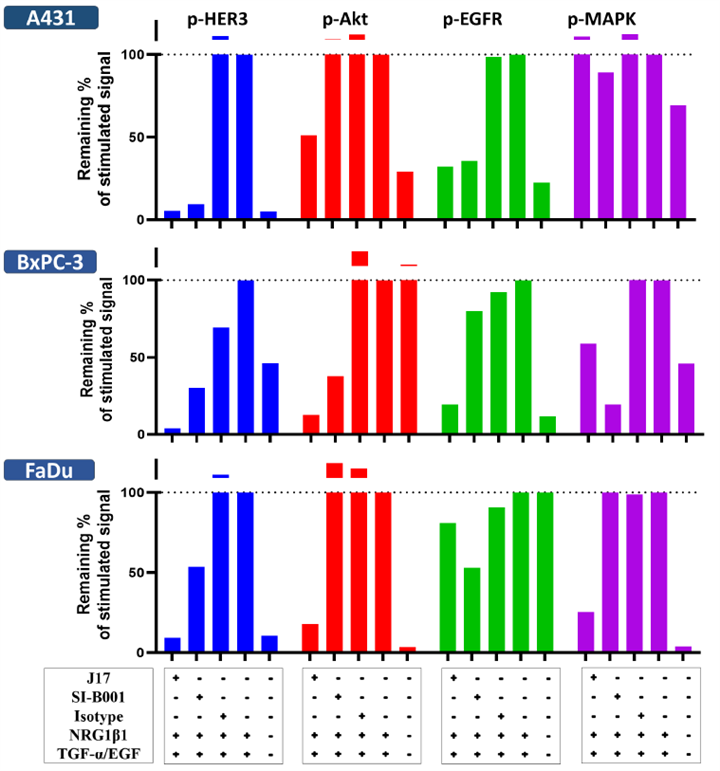
Notes:J17, CS5007’s antibody. SI-B001, the naked antibody of BL-B01D1, was synthesized based on published literature. The baseline signal was defined as the signal detected with the cell lysate harvested from the pre-stimulated tumor cells including BxPC-3, FaDu and A431.
3. Rapid and Deep Internalization
Using the Incucyte® Real-Time Live-Cell Imaging, the internalization profile of CS5007 was evaluated in A431, BxPC-3, FaDu, SW620 and MDA-MB-468 cells with pH sensor dye. CS5007 was efficiently and rapidly internalized across all tested tumor cell lines in a concentration-dependent manner and efficiently trafficked to the lysosome. Similar results were observed for the naked antibody J17. Notably, in SW620 cells with low EGFR expression, CS5007 maintains efficient drug delivery through the HER3-mediated internalization pathway.
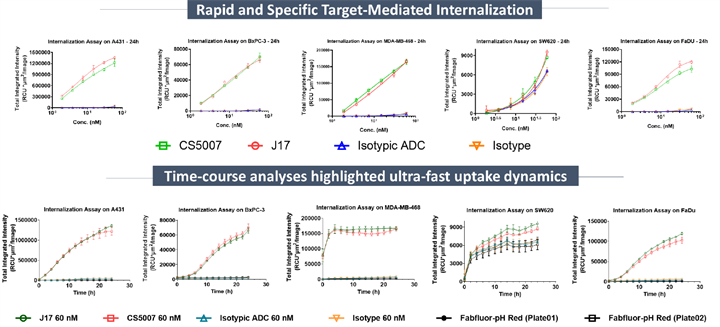
Notes:J17, CS5007’s antibody.
4. Potent and Broad-Spectrum In Vitro Anti-Tumor Activity
In vitro cytotoxicity was evaluated using CellTiter-Glo® (CTG) luminescent assay across 6 human tumor cell lines. CS5007 exhibits potent, nanomolar-level, antigen-dependent cell-killing activity across a broad spectrum of tumor cell lines, including non-small cell lung cancer (NSCLC), squamous cell carcinoma (SCC), colorectal cancer (CRC), squamous cell carcinoma (SCCHN), pancreatic cancer (PANC), and breast cancer (BC).
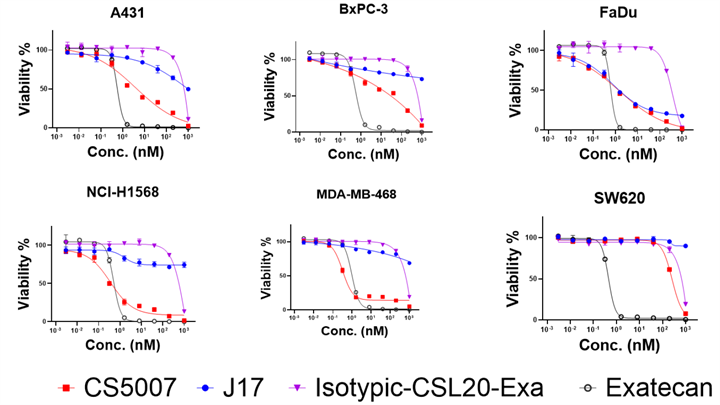
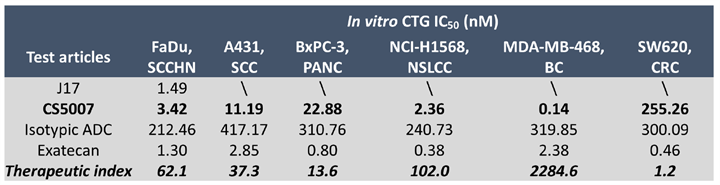
Notes: Therapeutic index (TI) is calculated as follows: TI=IC50 of Isotypic ADC / IC50 of CS5007
5. Significant Bystander Killing Effect
The bystander effect of CS5007 was assessed using a co-culture system of NCI-H1568 cells (antigen-positive, Ag+) and NCI-H524 cells (antigen-negative, Ag-) by CTG and flow cytometry (FCM) assays. In mono-culture systems, CS5007 induced cytotoxicity on H1568 cells, but not on HCI-H524 cells. In the co-culture system, CS5007 eliminated not only antigen-positive (NCI-H1568) tumor cells but also adjacent antigen-negative (NCI-H524) tumor cells, demonstrating an ability to address tumor heterogeneity and expand the therapeutic range.
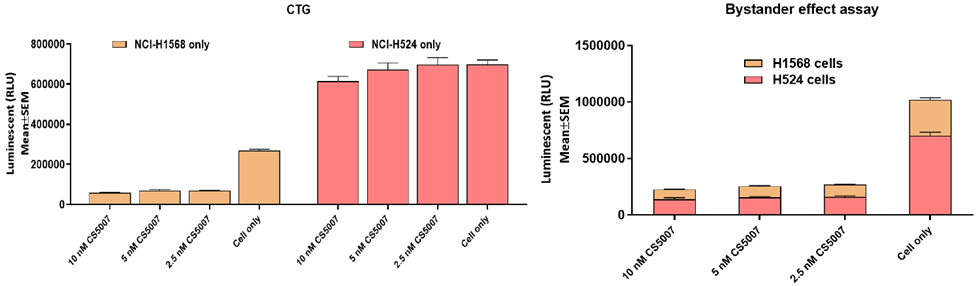
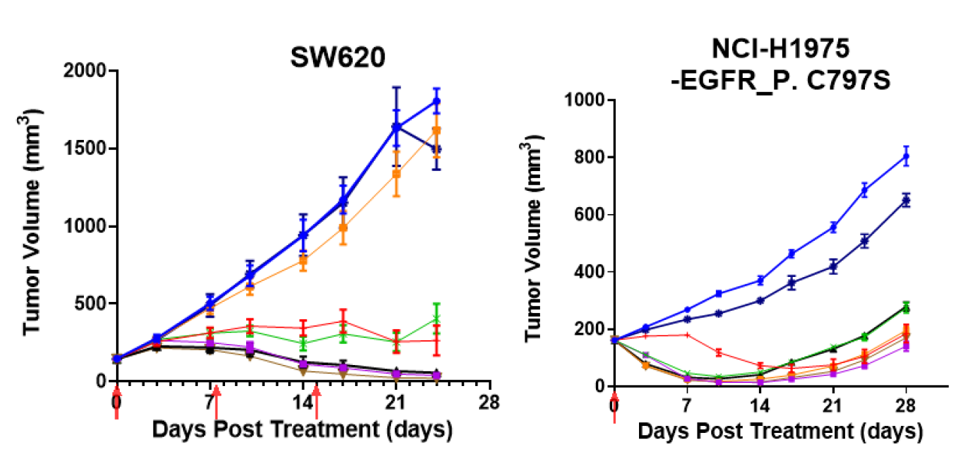
6. Broad-Spectrum In Vivo Anti-tumor Activity and Breakthrough in Resistant Models
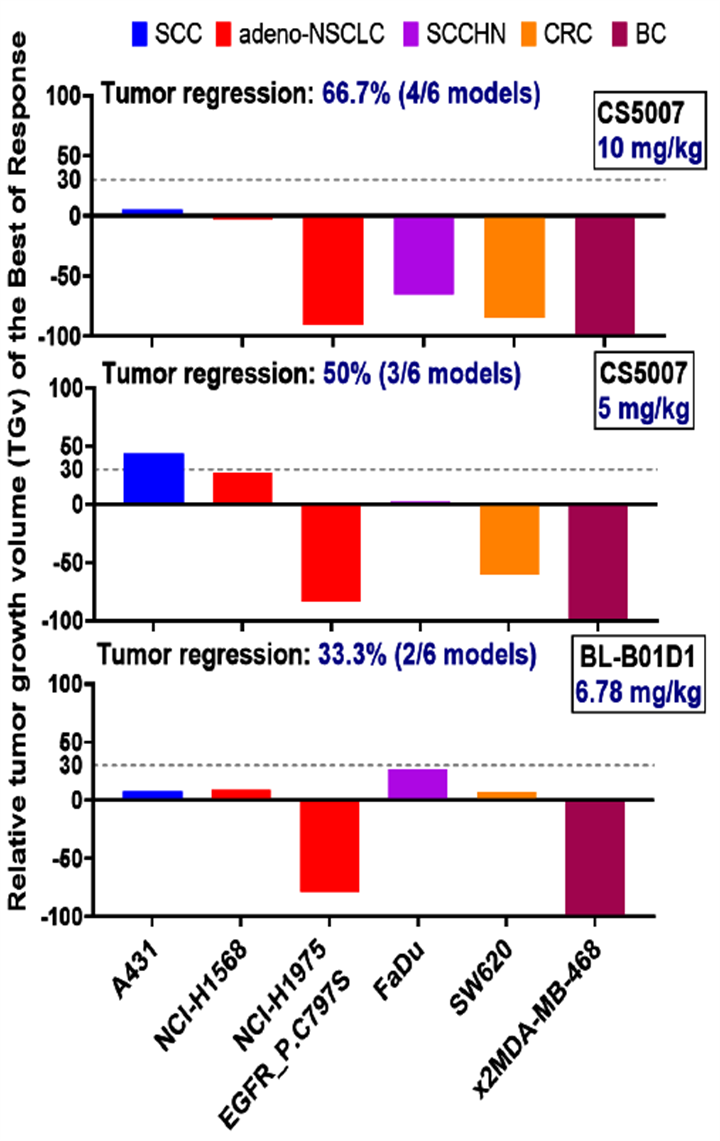
Notes: 1) J17, CS5007’s antibody. 2) BL-B01D1 is biosimilar synthesized by MCE. 3) Relative tumor growth volume (TGv, %) was calculated according to the following equation: TGv (%) = 100 × (Ti -T0)/T0, and if not, TGv (%) = 100 × (Ti -T0)/(Vi-V0). 4) The dose of BL-B01D1 (6.78 mg/kg) was determined as the dose level providing molar-equivalent payload carried by 10 mg/kg CS5007. With the exception on FaDu model, BL-B01D1 was given 5 mg/kg single dose. 5) the presented data is the summarized best of response after the 1st cycle treatment (7 days).
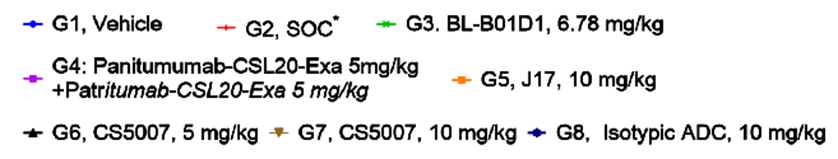
7. Favorable PK/PD Profile
CS5007 demonstrated a superior pharmacokinetics (PK) / Pharmacodynamics (PD) profile compared to BL-B01D1 in the FaDu CDX model:
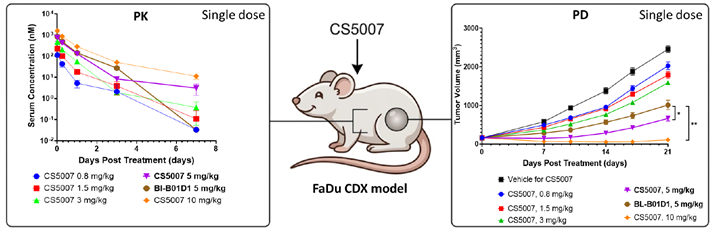

8. Favorable Safety and Tolerability
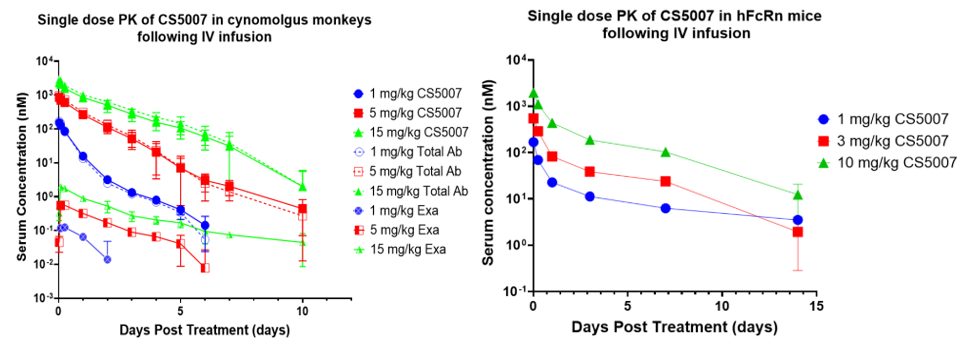
9. Phase I Clinical Trial Plan
CStone plans to initiate the Investigational New Drug (IND) application for CS5007 in the first half of 2026. The planned CS5007-101 study will be a monotherapy dose-escalation and expansion study designed to evaluate safety and recommended phase II dose (RP2D) in patients with advanced solid tumors. The study plans to enroll approximately 70 adult patients with advanced solid tumors who have progressed on or are ineligible for standard treatment or have no effective treatment options.
In summary, CS5007 is a highly promising bispecific ADC that demonstrates potent anti-tumor activity alongside a favorable safety and PK profile. Preclinical findings indicate that CS5007 binds with exceptional affinity, triggers rapid internalization across tumors with diverse EGFR and HER3 expression, effectively blocks dual downstream signaling pathways, and exhibits robust bystander-mediated tumor growth inhibition. These data provide a strong rationale to advance CS5007 into clinical investigation in solid tumors.
About CStone
CStone (HKEX: 2616), established in late 2015, is an innovation-driven biopharmaceutical company focused on the research and development of therapies for oncology, autoimmune/inflammation, and other key disease areas. Dedicated to addressing patients’ unmet medical needs in China and globally, the Company has made significant strides since its inception. To date, the Company has successfully launched 4 innovative drugs and secured approvals for 21 new drug applications covering 9 indications. The company’s pipeline is balanced by 16 promising candidates, featuring antibody-drug conjugates (ADCs), multispecific antibodies, immunotherapies and precision medicines. CStone also prides itself on a management team with comprehensive experiences and capabilities that span the entire drug development spectrum, from preclinical and translational research to clinical development, drug manufacturing, business development, and commercialization.
For more information about CStone, please visit: www.cstonepharma.com.
IR contact: ir@cstonepharma.com
PR contact: pr@cstonepharma.com
Forward-looking statements
The forward-looking statements made in this article only relate to events or information as of the date when the statements are made in this article. Except as required by law, we undertake no obligation to update or publicly revise any forward-looking statements, whether as a result of new information, future events or otherwise, after the date on which the statements are made or to reflect the occurrence of unanticipated events. You should read this article completely and with the understanding that our actual future results or performance may be materially different from what we expect. All statements in this article are made on the date of publication of this article and may change due to future developments.
Disclaimer: only for communication and scientific use by medical and health professionals, it is not intended for promotional purposes.
Your privacy is important for us. We use cookies to enhance your experience when visiting our websites: performance cookies show us how you use this website, functional cookies remember your preferences and targeting cookies help us to share content relevant to you. Select “Accept all” for giving your consent to all cookies or select “Reject all” for using only strictly necessary cookies.