Suzhou, China, April 20th, 2026—CStone Pharmaceuticals (“CStone,” HKEX: 2616), an innovation-driven biopharmaceutical company focused on the research and development of therapies for oncology, immunology, inflammation, and other key disease areas, today announced that the Company presented the latest preclinical data for three proprietary pipeline assets at the American Association for Cancer Research (AACR) Annual Meeting (from April 17 to 22), including CS5007 (EGFR/HER3 ADC), CS5006 (ITGB4 ADC), and CS5008 (DLL3/SSTR2 ADC).
CStone's Proprietary ADC Technology Platform
All three antibody-drug conjugates (ADCs) presented at AACR – CS5007 (EGFR/HER3 ADC), CS5006 (ITGB4 ADC), and CS5008 (DLL3/SSTR2 ADC) – are developed utilizing CStone’s proprietary ADC technology platform, which incorporates the following core features:
CS5008 – SSTR2/DLL3 Bispecific ADC
DLL3 and SSTR2 are both highly overexpressed in small cell lung cancer (SCLC) and neuroendocrine tumors/carcinomas (NETs/NECs). DLL3 is overexpressed in more than 70% of SCLC and 64% of NECs patients, while SSTR2 is overexpressed in over 50% of SCLC and approximately 90% of G1/G2 NETs patients. Therefore, dual targeting of SSTR2 and DLL3 holds great promise for overcoming intra- and inter-tumoral heterogeneity in SCLC and NETs/NECs, potentially improving efficacy and broadening the addressable patient population. Despite initial high sensitivity to chemotherapy and/or radiotherapy, most SCLC patients develop therapeutic resistance within one year. Accumulating evidence highlights that the heterogeneity and plasticity of SCLC are closely associated with the development of distant metastases and chemoresistance, which remain major obstacles to improving clinical outcomes.
CS5008 is a bispecific ADC constructed with: 1) an anti-DLL3 and SSTR2 human IgG1 antibody; 2) CStone proprietary hydrophilic CSL20 linker; 3) exatecan (Exa) as payload, conjugated with a DAR of approximately 4.
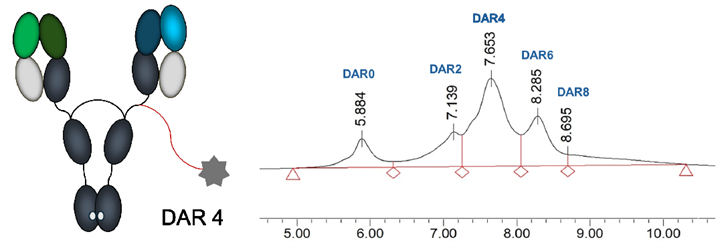
*Beta-glucuronidase exclusively functions within cells and is highly expressed in tumor cells.
Key Highlights:
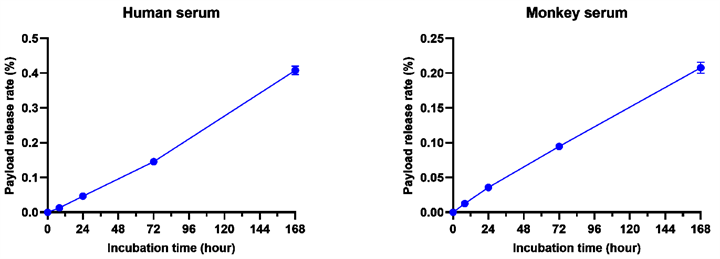
1. Excellent In Vitro Serum Stability
CS5008 demonstrated excellent stability in both human and cynomolgus monkey serum. After 7 days of incubation at 37°C, the toxin release rate was below 0.5%.
2. Rapid and Deep Internalization
CS5008 triggers rapid and deep internalization on SSTR2- and/or DLL3-positive tumor cells.
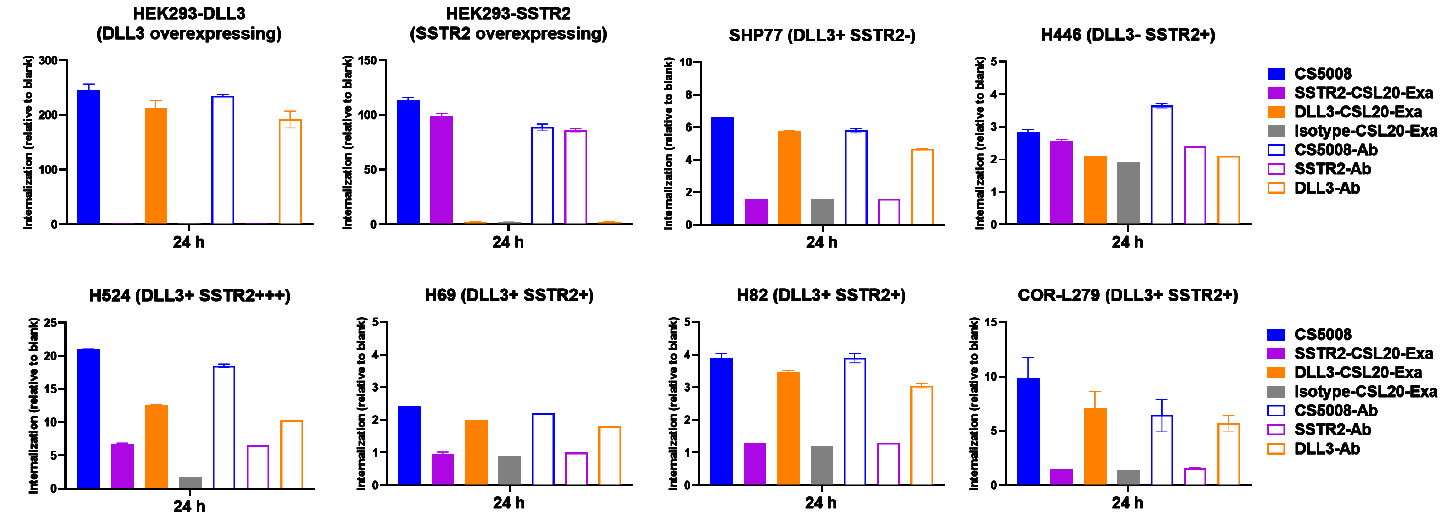
Note: SSTR2-Ab and DLL3-Ab were the parental clones applied to form CS5008 bsAb.
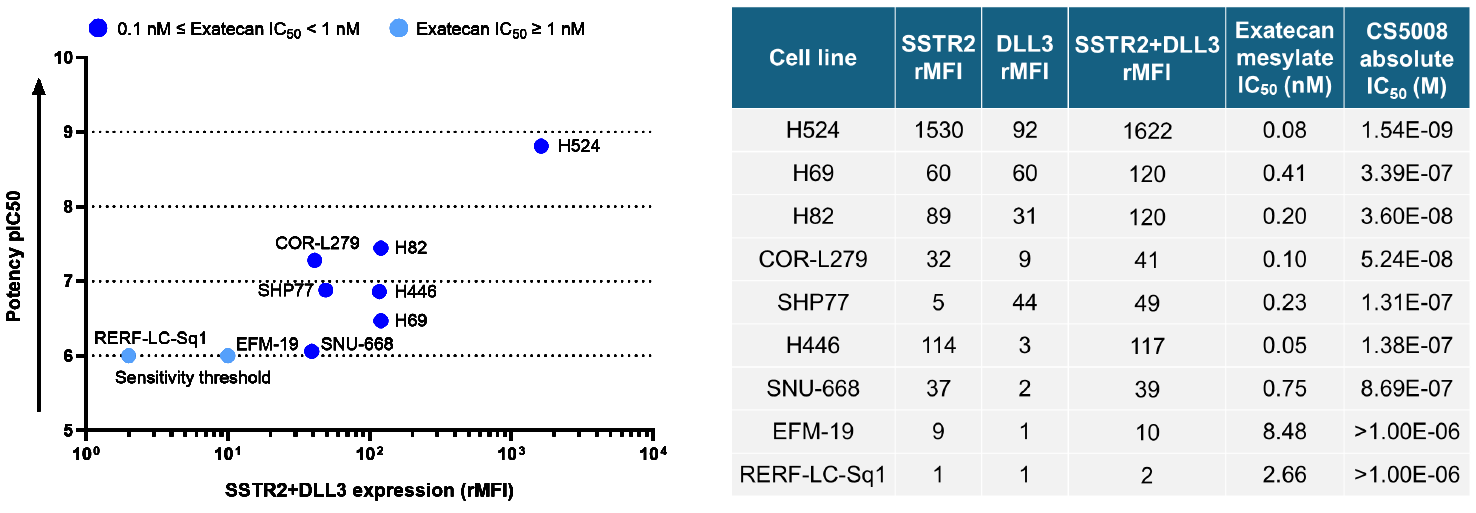
3. Potent and Antigen-Dependent In Vitro Killing Activity
CS5008 efficiently and specifically kills tumor cells expressing DLL3 or SSTR2 antigens, and the killing effect is significantly positively correlated with the expression level of the antigens (SSTR2+DLL3).
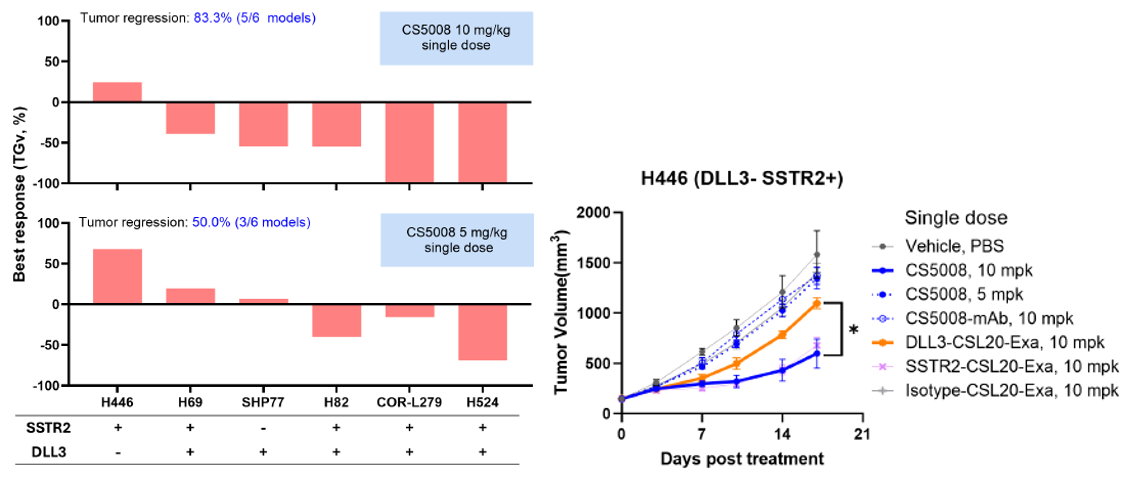
4. Broad-Spectrum and Potent In Vivo Anti-Tumor Activity
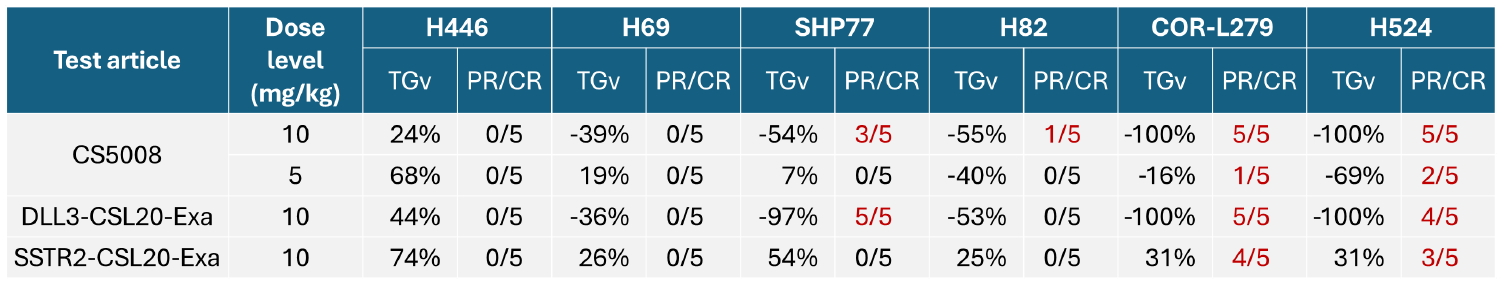
In SCLC CDX models, CS5008 demonstrated potent and broad-spectrum anti-tumor effects. A single dose induced tumor regression in various tumor models with different antigen expression levels. Notably, CS5008 outperformed DLL3-CSL20-Exa in the H446 (DLL3 negative SCLC) CDX model, indicating its potential to overcome tumor heterogeneity.
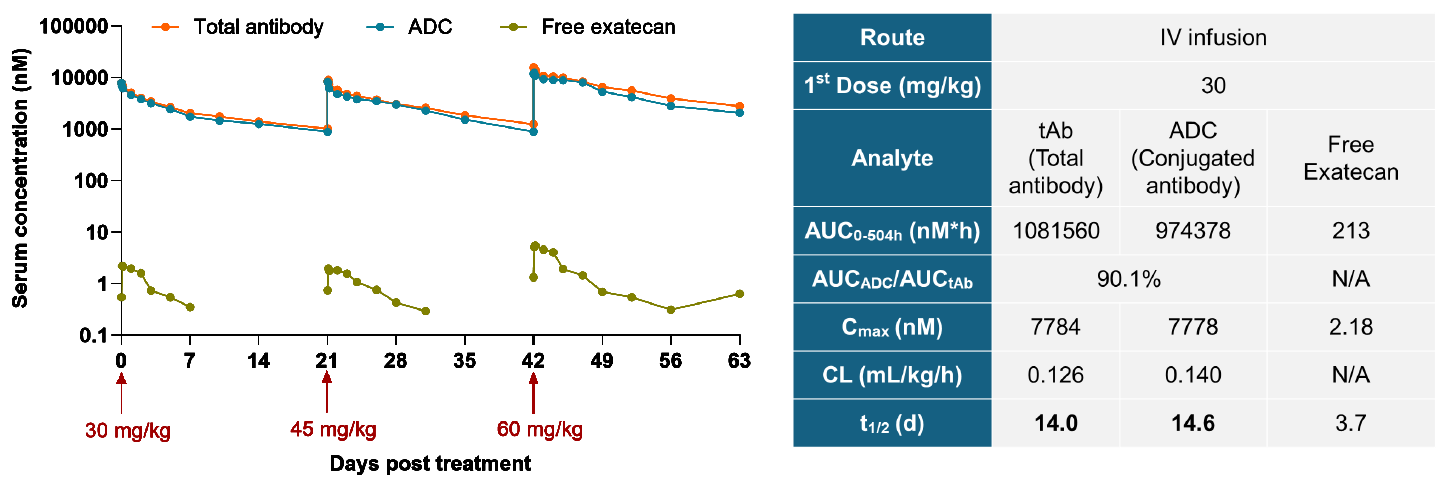
5. Favorable PK and Safety Profile
CS5008 exhibited a favorable PK profile in cynomolgus monkeys:

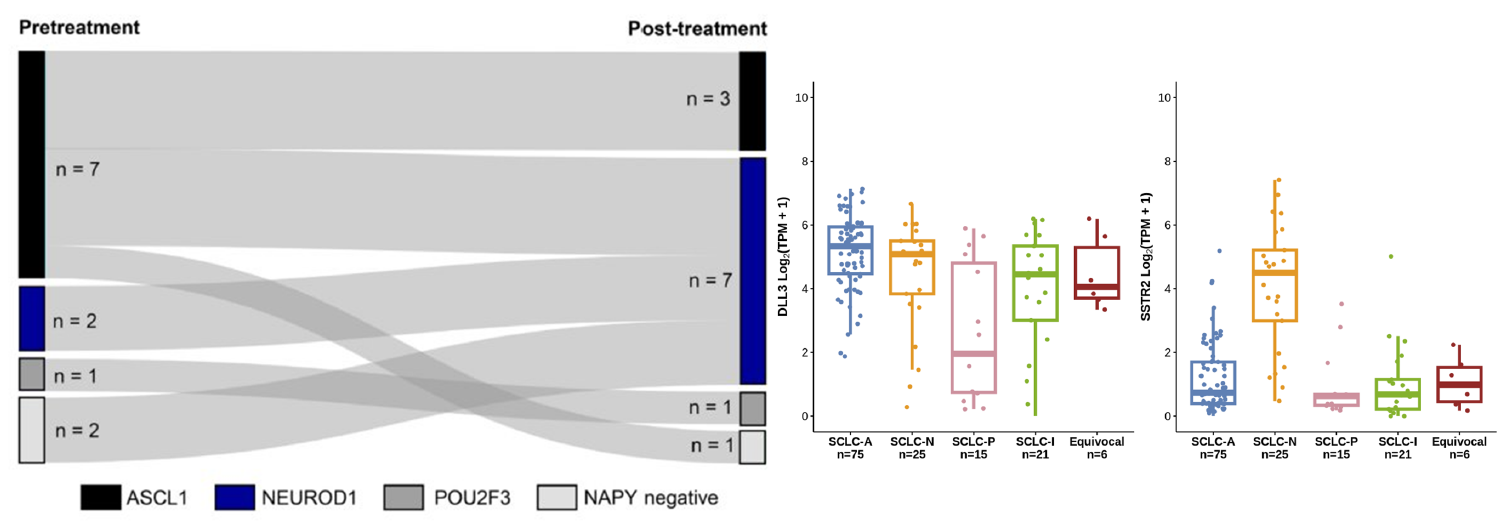
6. Potential New Strategy to Overcome Subtype Switching Resistance
By simultaneously targeting both antigens, CS5008 can effectively address the resistance challenge induced by treatment-driven molecular subtype switching in SCLC (e.g., from SCLC-A to SCLC-N).*
Notes. *: Yoshida T. WCLC, 2025; 1: Chiang CL et al. Lung Cancer. 2024 Feb; 2: Heeke S et al. Cancer Cell. 2024 Feb
CS5008 is a highly promising bispecific ADC with broad-spectrum, potent anti-tumor activity and favorable safety and PK profiles. Preclinical studies demonstrate that via rapid and deep internalization, CS5008 enables efficient and specific killing of tumor cells with varying DLL3 and SSTR2 expression and exhibits broad, robust in vivo anti-tumor activity in xenograft models. In NHPs, CS5008 displays favorable tolerability and excellent PK properties. Furthermore, CS5008 features high antibody yield, strong ADC stability, and good developability. Collectively, these data support advancing CS5008 into IND-enabling studies and subsequent clinical evaluation for solid tumors including SCLC, NECs, etc.
CStone plans to initiate the IND application for CS5008 in the second half of 2026.
About CStone
CStone (HKEX: 2616), established in late 2015, is an innovation-driven biopharmaceutical company focused on the research and development of therapies for oncology, autoimmune/inflammation, and other key disease areas. Dedicated to addressing patients’ unmet medical needs in China and globally, the Company has made significant strides since its inception. To date, the Company has successfully launched 4 innovative drugs and secured approvals for 21 new drug applications covering 9 indications. The company’s pipeline is balanced by 16 promising candidates, featuring antibody-drug conjugates (ADCs), multispecific antibodies, immunotherapies and precision medicines. CStone also prides itself on a management team with comprehensive experiences and capabilities that span the entire drug development spectrum, from preclinical and translational research to clinical development, drug manufacturing, business development, and commercialization.
For more information about CStone, please visit: www.cstonepharma.com.
IR contact: ir@cstonepharma.com
PR contact: pr@cstonepharma.com
Forward-looking statements
The forward-looking statements made in this article only relate to events or information as of the date when the statements are made in this article. Except as required by law, we undertake no obligation to update or publicly revise any forward-looking statements, whether as a result of new information, future events or otherwise, after the date on which the statements are made or to reflect the occurrence of unanticipated events. You should read this article completely and with the understanding that our actual future results or performance may be materially different from what we expect. All statements in this article are made on the date of publication of this article and may change due to future developments.
Disclaimer: only for communication and scientific use by medical and health professionals, it is not intended for promotional purposes.
Your privacy is important for us. We use cookies to enhance your experience when visiting our websites: performance cookies show us how you use this website, functional cookies remember your preferences and targeting cookies help us to share content relevant to you. Select “Accept all” for giving your consent to all cookies or select “Reject all” for using only strictly necessary cookies.